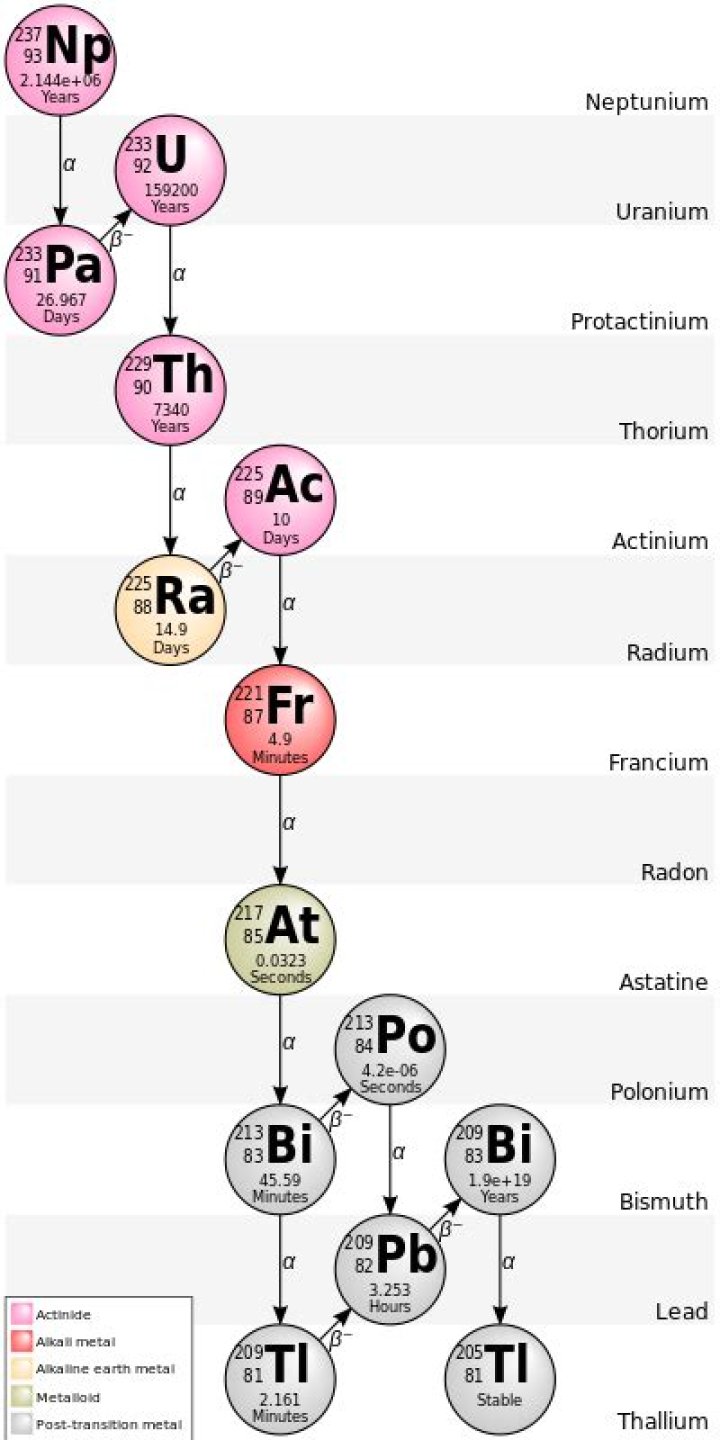
It has 83 protons and a magic number of 126 neutrons, and an atomic mass of 208.9803987 amu (atomic mass units)….Bismuth-209.
| General | |
|---|---|
| Natural abundance | 100% |
| Half-life | 2.01×1019 years |
| Parent isotopes | 209Pb (β−) 209Po (β+) 213At (α) |
| Decay products | 205Tl |
What is the half-life of PB 212?
10.6 hours
Lead-212 has a half-life of 10.6 hours. Its decay chain includes the short-lived isotopes bismuth-212, polonium-212 and thallium-208, which all emit either alpha or beta during decay over about another hour.
How do you find the half-life of bismuth?
The half-life was calculated to be (1.9 +/- 0.2 ) x 1019 years, which is in good agreement with the theoretical prediction of 4.6 x 1019 years. The technique could be also be used to accurately detect beta and gamma decays.
What is the half-life of bismuth?
about 20,000,000,000,000,000,000 years
But although bismuth is no longer the paragon of stability, it’s no flake either. The researchers found that the element has what may be the longest half-life of any isotope: about 20,000,000,000,000,000,000 years.
What element has the longest life?
The data helped the collaboration make the first definitive measurement of xenon-124’s half-life: 18 billion trillion years. “This is the longest lifetime that we have ever directly measured.”
How fast does bismuth decay?
A mole of any substance is composed of Avogadro’s number, 6.022×1023 of constituents. Dividing this by 209, we find our sample contains 2.9×1021 atoms of bismuth. Using the formulæ above, we find that 105 atoms of bismuth in our sample will decay, on average, every year, or around one every three and a half days.
Is lead 212 dangerous?
Radio-toxicity for Lead-212 is HIGH (from scale of very high-very low). Lead-212 is used commonly to treat melanoma, breast cancer, and ovarian cancer.
What product nuclei are formed when 208 Po undergoes alpha decay?
Explanation: Polonium-212 undergoes alpha decay to produce lead-208, which means that it gives off alpha radiation, i.e. alpha particles.
What is the longest radioactive half-life?
The data helped the collaboration make the first definitive measurement of xenon-124’s half-life: 18 billion trillion years. “This is the longest lifetime that we have ever directly measured.” This decay process is called two-neutrino double electron capture.
What material has the longest half-life?
The half-life of xenon-124 — that is, the average time required for a group of xenon-124 atoms to diminish by half — is about 18 sextillion years (1.8 x 10^22 years), roughly 1 trillion times the current age of the universe. This marks the single longest half-life ever directly measured in a lab, Wittweg added.
What is the normal phase of bismuth?
Solid
| Name | Bismuth |
|---|---|
| Melting Point | 271.3° C |
| Boiling Point | 1560.0° C |
| Density | 9.8 grams per cubic centimeter |
| Normal Phase | Solid |
Why are elements bigger than bismuth radioactive?
Nuclei at or just below a magic number will be more tightly bound than nuclei just above the magic numbers. Thus you’ll often find more radioactive nuclei just above a magic number: because they are less tightly bound than the magic nuclei, and thus have a larger mass difference.
Are there any long lived isotopes of bismuth?
Bismuth has no stable isotopes, but does have one very long-lived isotope; thus, the standard atomic weight can be given as 208.98040(1).
What is the standard atomic weight of bismuth?
Bismuth has no stable isotopes, but does have one very long-lived isotope; thus, the standard atomic weight can be given as 208.980 40(1).
How is bismuth 213 used to treat leukemia?
Commercially, the isotope bismuth-213 can be produced by bombarding radium with bremsstrahlung photons from a linear particle accelerator. In 1997, an antibody conjugate with 213 Bi (which has a 45-minute half-life, and decays with the emission of an alpha particle) was used to treat patients with leukemia.
Where is bismuth 213 found in the decay chain?
Bismuth-213 is also found in the decay chain of uranium-233, which is the fuel “bred” by thorium reactors . ^ mBi – Excited nuclear isomer.
What element has the longest half life?
nvm problm solved: I finally found the element with longest half life, as stated in my element book,” Uranium is the last and heaviest of the natural elements” (203). “its half-life of 4.6 billion years makes it the longest-lived of all isotopes.
What is the formula for calculating half life?
2k
How do you solve half life problems?
Multiply both sides by t1/2{\\displaystyle t_{1/2}} and divide both sides by the entire left side to solve for half-life. Since there are logarithms in the final expression, you’ll probably need a calculator to solve half-life problems.
How long can bismuth last?
But although bismuth is no longer the paragon of stability, it’s no flake either. The researchers found that the element has what may be the longest half-life of any isotope: about 20,000,000,000,000,000,000 years.