This is a list of radioactive nuclides (sometimes also called isotopes ), ordered by half-life from shortest to longest, in seconds, minutes, hours, days, and years. Current methods make it difficult to measure half-lives between approximately 10 -19 and 10 -10 seconds.
Are there any radioactive elements with a half life of 1000 years?
“Barely Radioactive Elements”. Retrieved 2018-03-09. Radioactive isotope table “lists ALL radioactive nuclei with a half-life greater than 1000 years”, incorporated in the list above.
What do you call the half life of a sample?
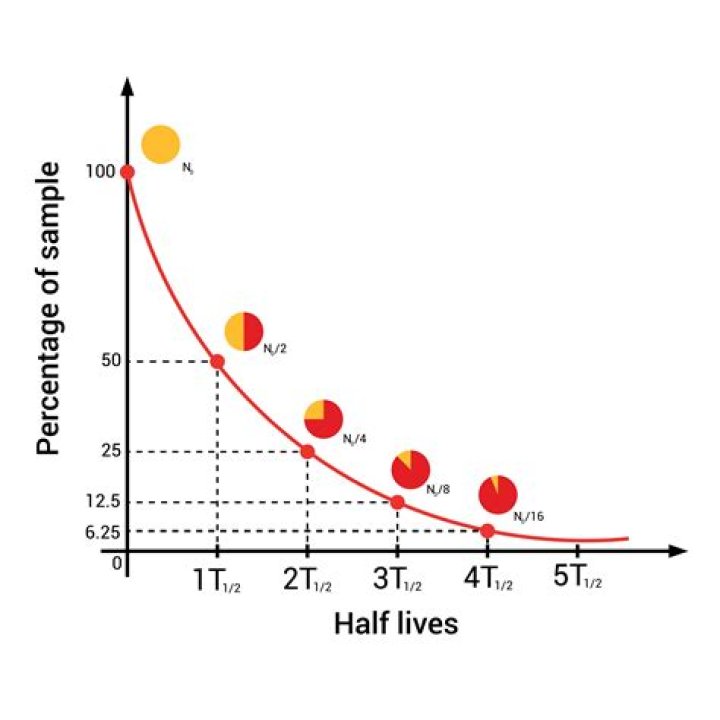
This is called the half-life. curriculum-key-fact. Half-life is the time it takes for half of the unstable nuclei in a sample to decay or for the activity of the sample to halve or for the count rate to halve. Count-rate is the number of decays recorded each second by a detector, such as the Geiger-Muller tube.
What is the half life of radioactive carbon 14?
The half-life of radioactive carbon-14 is 5,730 years. If a sample of a tree (for example) contains 64 grams (g) of radioactive carbon after 5,730 years it will contain 32 g, after another 5,730 years that will have halved again to 16 g.
What is the half life of a radioisotope?
If a radioisotope has a half-life of 14 days, half of its atoms will have decayed within 14 days. In 14 more days, half of that remaining half will decay, and so on.
How is the half life of a radioactive substance measured?
Half-Life The half-life of a radioactive substance is a characteristic constant. It measures the time it takes for a given amount of the substance to become reduced by half as a consequence of decay, and therefore, the emission of radiation.
What is meant by half life in physics?
It’s a Question of Physics: What is meant by half-life? The half-life of a radioactive substance is a characteristic constant. It measures the time it takes for a given amount of the substance to become reduced by half as a consequence of decay, and therefore, the emission of radiation.
Is there a half life for radioactive iodine?
Not exactly. Every half life the amount of radioactive iodine is reduced by 50%. The other 50% is no longer I131, but is now stable (non-radioactive Xenon 131. Here’s an example: Day #24 – 12.5 millicuries remains, etc.