What are the uses of Neutralisation in everyday life?
Sodium hydroxide: used in the production of soaps, paper, and synthetic fibres. Magnesium hydroxide: used as an antacid to neutralise the acid in the stomach. Ammonium hydroxide: manufacturing of fertilisers. Calcium hydroxide: used for manufacturing bleaching powder.
What are the products of Neutralisation?
In the neutralisation reaction between an acid and a metal carbonate, there are three products. The hydrogen ions (H +) from the acid react with the carbonate ions (CO 3 2-) to form water and carbon dioxide gas. A salt is also produced.
What is Neutralisation example?
Hint: The neutralization reaction is the one in which an acid reacts with an equimolar amount of base to give salt and water. The example could be a reaction between any strong acid and a base. The sodium chloride formed is a result of neutralization reaction.
What is importance of neutralization?
A neutralization reaction can be important for many reasons. One example is in agriculture. Neutralization reactions are when an acid and base react, typically forming water and salt. These processes will help return pH levels to more neutral levels.
What is Neutralisation explain with examples?
What is Neutralisation with example?
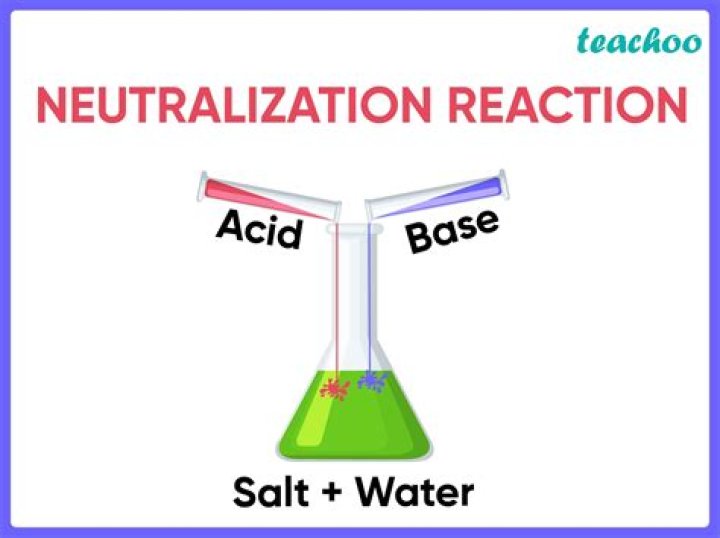
A neutralization reaction is when an acid and a base react to form water and salt and involves the combination of hydrogen ions and hydroxyl ions to generate water. The neutralization of a strong acid and strong base has a pH equal to 7. Example – 1: When Sodium hydroxide is added to hydrochloric acid.
Is hydrochloric acid a reactant?
For instance, in the equation HCl + NaOH → NaCL + H2O, the HCl (hydrochloric acid, a strong acid) and NaOH (sodium hydroxide, a strong base) are the reactants.
What is Neutralisation answer?
Definition: Neutralisation is a reaction where an acid reacts with an alkali to form a neutral solution of a salt and water.
What is neutralization reaction simple definition?
Definitions of neutralization reaction. a chemical reaction in which an acid and a base interact with the formation of a salt; with strong acids and bases the essential reaction is the combination of hydrogen ions with hydroxyl ions to form water.
What is Neutralisation in science?
Neutralisation is the reaction between an acid and a base. Acids react with metals, bases and carbonates to produce salts. Combined Science.
When to use neutralization in a chemical reaction?
Neutralization is used in many applications. In the context of a chemical reaction the term neutralization is used for a reaction between an acid and a base or alkali. Historically, this reaction was represented as
Why is neutralisation important in our daily life?
Neutralisation in our daily life plays an extremely important role. Let’s find out. Let us look at some common neutralisation reactions in daily life that we experience on a daily basis and understand the importance of neutralisation reactions in our daily life.
Which is the best example of neutralization in food?
If a strong acid and strong base react it gives salt of neutral nature. similarly strong base and a week acid react the salt is basic in nature. 1. Tooth decay or cavities Chocolates and decaying food particles produce acid in our mouth which reacts with enamel i.e. calcium phosphate and leads to cavities.
Which is an example of a neutralization titration?
Neutralization is used in many applications. Animation of a strong acid–strong base neutralization titration (using phenolphthalein ). The equivalence point is marked in red.
Neutralization is used in many applications. In the context of a chemical reaction the term neutralization is used for a reaction between an acid and a base or alkali. Historically, this reaction was represented as
Neutralisation in our daily life plays an extremely important role. Let’s find out. Let us look at some common neutralisation reactions in daily life that we experience on a daily basis and understand the importance of neutralisation reactions in our daily life.
If a strong acid and strong base react it gives salt of neutral nature. similarly strong base and a week acid react the salt is basic in nature. 1. Tooth decay or cavities Chocolates and decaying food particles produce acid in our mouth which reacts with enamel i.e. calcium phosphate and leads to cavities.
Neutralization is used in many applications. Animation of a strong acid–strong base neutralization titration (using phenolphthalein ). The equivalence point is marked in red.
What is an example of Neutralisation in everyday life?
Neutralization in everyday life For example; during indigestion, taking milk of magnesia (magnesium hydroxide) gives us relief as it neutralizes the effect of excess acid produced inside the stomach. Vinegar is used to cure wasp stings that are alkaline in nature.
Where is Neutralisation useful?
Neutralisation is the reaction of an acid with a base that results in the pH moving towards seven. It is a useful process that occurs in everyday life such as in the treatment of acid indigestion and the treating of acidic soil by adding lime.
Uses of Bases in Daily Life: Sodium hydroxide: used in the production of soaps, paper, and synthetic fibres. Magnesium hydroxide: used as an antacid to neutralise the acid in the stomach. Ammonium hydroxide: manufacturing of fertilisers. Calcium hydroxide: used for manufacturing bleaching powder.
Why do we need Neutralisation?
A neutralization reaction can be important for many reasons. Neutralization reactions are when an acid and base react, typically forming water and salt. These processes will help return pH levels to more neutral levels. Soil can sometimes become more acidic (under 5.5).
What is neutralization example?
When a strong acid reacts with a strong base the resultant salt is neither acidic nor basic in nature i.e. it is neutral. For example when HCl (Hydrochloric acid), a strong acid, reacts with NaOH, a strong base, then the resulting salt is sodium chloride and water.
How does Neutralisation reaction play an important role in our daily life?
Following are some neutralization reactions that we observe in our daily life: In the treatment of ant sting: Some people are highly allergic to ant sting as it releases formic acid. In the treatment of indigestion: Hydrochloric acid secreted in the stomach helps in the digestion of food.
How is Neutralisation used in toothpaste?
Neutralisation is an important chemical reaction. It occurs when an acid is mixed with an alkali until a neutral solution is obtained. However, if we brush our teeth with toothpaste, the alkaline in the toothpaste will neutralise the acids produced by the bacteria in our mouth. Tooth decay can be prevented.
What is the importance of neutralization in our daily life?
What is the neutralization formula?
The overall equation for this reaction is: NaOH + HCl → H2O and NaCl. Now let’s break this reaction down into two parts to see how each product forms.
What is Neutralisation give example?
How does neutralization play an important role in our life give three examples to explain?
Question: Neutralization reactions play an important role in our daily life. In the treatment of soil: Slake lime or quick lime is used to neutralize the acidity level of the soil. if the soil is too basic fertilizers made from vegetables and fruits which are acidic in nature are added to the soil.
What is Neutralisation explain with example?
The reaction between an acid and bases is called a neutralization reaction. The result of the reaction is salt and water. Acid + Base → Salt + Water + heat. For example, when hydrochloric acid(acid) and sodium hydroxide(base) react they form sodium chloride(salt) and water. Also, heat is evolved.