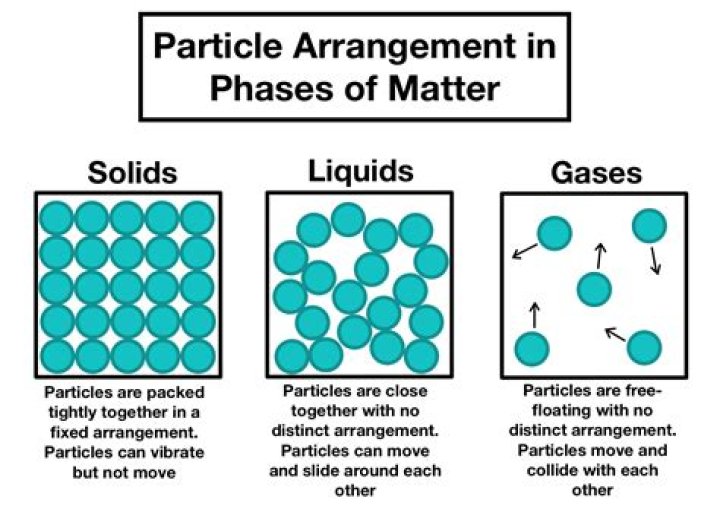
specific heat. Solids that are made up of particles arranged in repeating geometric patterns are called (blank) Crystalline. When the crystal structure of matter begins to collapse into a swarm of freely flowing molecules, the matter is moving from the (blank) state to the (blank) state.
When does matter move from a solid to a liquid?
Solids that are made up of particles arranged in repeating geometric patterns are called (blank) Crystalline. When the crystal structure of matter begins to collapse into a swarm of freely flowing molecules, the matter is moving from the (blank) state to the (blank) state. solid, liquid.
How are amorphous particles arranged in a crystal?
Amorphous-particles are randomly oriented. If the atoms or molecules are uniquely arranged in crystalline solid or liquid we call it as a crystal structure. A crystal posses long range order and symmetry. The main property of crystal structure is its periodicity. This periodicity is due to the arrangement of atoms/molecules in the lattice points.
How is the appearance of a crystal related to its internal arrangement?
However, these are not entirely independent since the external appearance of a crystal is often related to the internal arrangement. For example, crystals of cubic rock salt (NaCl) are physically cubic in appearance.
Can an amorphous solid transition to a crystalline state?
Also, amorphous solids may undergo a transition to the crystalline state under appropriate conditions. Figure 2. (a) Diboron trioxide, B2O3, is normally found as a white, amorphous solid (a glass), which has a high degree of disorder in its structure.
Which is a characteristic of the solid state of matter?
All exhibit high thermal and electrical conductivity, metallic luster, and malleability. Many are very hard and quite strong. Because of their malleability (the ability to deform under pressure or hammering), they do not shatter and, therefore, make useful construction materials. The melting points of the metals vary widely.
How are metallic solids formed in the solid state?
Metallic solids such as crystals of copper, aluminum, and iron are formed by metal atoms Figure 4. The structure of metallic crystals is often described as a uniform distribution of atomic nuclei within a “sea” of delocalized electrons.
When is constructive interference observed in a crystal?
Constructive interference is observed when the path difference between the two reflected beams in (a) = n. The path difference in (a) is 2my. Since my/d = sin 2my = 2dsin n where d is the interplanar spacing. 2d (1,0,0)sin This is a first order reflection 2d (1,0,0)sin 2 This is a secondorder reflection