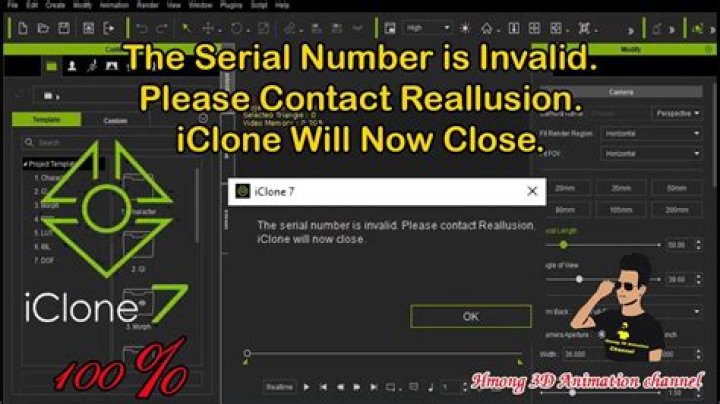
Rated 5 stars based on 1 vote. Sorry to seem incredible think but where do i enter the serial number? my 15 day trial has run out according to the software. So I have purchased the full pro license and registered it online.
What is a serial number and what is it for?
A serial number is a unique, identifying number or group of numbers and letters assigned to an individual piece of hardware or software. Other things have serial numbers as well, though, including banknotes and other similar documents. The idea behind serial numbers…
Where is the serial number on an Australian Lithgow?
They may or may not have the serial stamped into the front part of the fore-end and in the case of the Australian Lithgow’s it may be stamped into the butt as well. When reference is made to an “all matching” SMLE the serial numbers must all be the same.
Where can I Find my product serial number?
Only an administrator can assign you as a Named User or End User and give you permissions to download and activate the software. When students, educators, and educational institutions download products from the Education Community , the product serial numbers are found in the following locations:
How to create a clinical trial registry identifier?
Definition: If a Secondary ID Type of “Other Grant/Funding Number,” “Registry Identifier,” or “Other Identifier” is selected, provide the name of the funding organization, clinical trial registry, or organization that issued the identifier. Limit: 119 characters.
When to submit ClinicalTrials.gov protocol Registration data?
ClinicalTrials.gov Protocol Registration Data Element Definitions for Interventional and Observational Studies March 7, 2019 This document describes the definitions for protocol registration data elements submitted to ClinicalTrials.gov for interventional studies (clinical trials) and observational studies.
What does verification date mean on ClinicalTrials.gov?
Record Verification Date * Definition: The date on which the responsible party last verified the clinical study information in the entire ClinicalTrials.gov record for the clinical study, even if no additional or updated information is being submitted. Overall Recruitment Status *