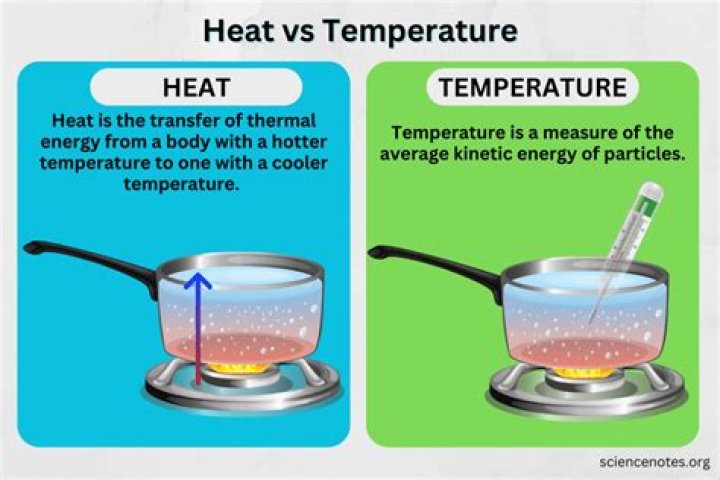
Which statement is true about the flow of heat energy heat energy?
Heat energy will always be transferred from the hotter to the cooler object. The objects will exchange thermal energy, until thermal equilibrium is reached, i.e. until their temperatures are equal. We say that heat flows from the hotter to the cooler object.
What is true about heat and work?
When energy is exchanged between thermodynamic systems by thermal interaction, the transfer of energy is called heat. Heat and work are related: work can be completely converted into heat, but the reverse is not true: heat cannot be completely converted to work.
Which statement is the best scientific definition for heat?
The term heat is defined as the internal energy of a substance. The change in internal energy of a system can be found by combining the heat energy added to a system minus the work done by the system.
Can radiation occur in vacuum?
Radiation can occur not only from solid surfaces but also from liquids and gases. While the transfer of energy by conduction or convection requires the presence of material medium, radiation does not. In fact, radiation transfer occurs most efficiently in vacuum.
What are the 5 sources of heat?
Here are only some of your many choices for heating energy sources: natural gas, propane (LP), oil, coal, wood, electricity, heat pumps, ground source heat pumps and solar energy.
What’s a fact about heat?
Heat is actually a form of energy. As atoms and molecules inside of an object move, they create heat energy. The faster they move, the hotter the object. Cold objects just have less heat energy than warmer objects, meaning their atoms and molecules are moving more slowly.
What is heat energy formula?
The equation for calculating heat energy is q=mCpΔT, where q is the heat variable, m is the mass of the object, Cp is the specific heat constant and ΔT is the temperature change.
Is it true that when object releases heat its energy increases?
Answer: True. Explanation: When the temperature of an object increases, the average kinetic energy of its particles increases. When the average kinetic energy of its particles increases, the object’s thermal energy increases.
Is heat absorbed positive or negative?
When heat is absorbed by the solution, q for the solution has a positive value. This means that the reaction produces heat for the solution to absorb and q for the reaction is negative. When heat is absorbed from the solution q for the solution has a negative value.
What is the relationship between energy work and heat?
Heat and work are two different ways of transferring energy from one system to another. The the distinction between Heat and Work is important in the field of thermodynamics. Heat is the transfer of thermal energy between systems, while work is the transfer of mechanical energy between two systems.
What best defines heat?
The study of the relationship between chemical reactions and changes in energy The energy required to move an object through a given distance The chemical equation of a reaction that includes energy as a reactant or a product The energy transferred between objects because of a difference in their temperature.
What is a fancy name for heat?
The condition or quality of being hot. warmth. fieriness. hotness. swelter.
Is a good absorber also a good emitter of radiation?
An object that is good at absorbing radiation is also a good emitter , so a perfect black body would be the best possible emitter of radiation. There are no known objects that are perfect at absorbing or emitting all radiation of every possible frequency that may be directed at it.
Does heat transfer in vacuum?
Yes. In vacuum heat transfer occurs through radiation mode of heat transfer. In vacuum heat can’t be transferred by conduction or convection.
What are 2 sources of heat?
Heating Energy Sources
- Gas Fired Water Heater. Here at Radiantec we often recommend the use of domestic water heaters instead of expensive boilers.
- Oil Fired Boilers or Water Heaters.
- Wood or Coal Heaters.
- Solar Collectors.
- Radiant Retrofit.
- Electric Heat.
- Geothermal.
What is the best source of heat?
Most of the time, natural gas is the most efficient means of heating the home, which is why most American households use it. Household Heating Systems: Although several different types of fuels are available to heat our homes, nearly half of us use natural gas. | Source: Buildings Energy Data Book 2011, 2.1.