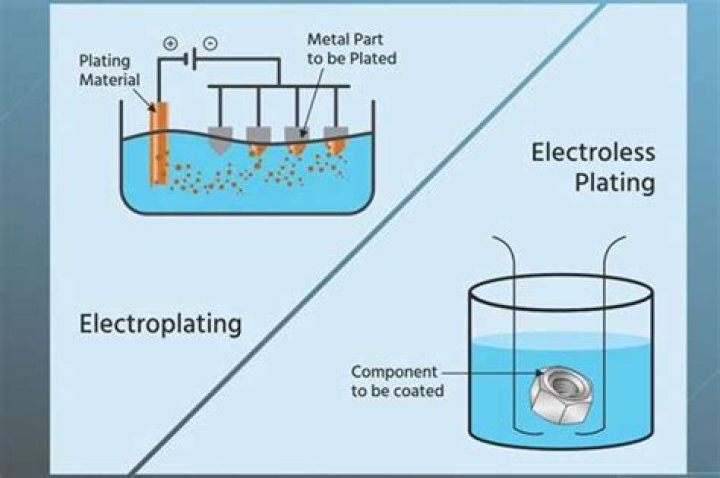
Improving properties One use of electroplating is to improve the resistance of metal objects to corrosion . It also improves their appearance and may be used to produce gold-plated jewellery. the positively charged anode is the plating metal. the electrolyte contains ions of the plating metal.
What is the main purpose of electroplating metals?
Electroplating is the process of coating surfaces with a metal coating using electricity. Electroplating creates a protective barrier to reduce friction and prevent tarnishing of a surface as well as protecting surfaces from wear and tear by applying a thin, durable metal coating.
What is electroplating and why it is done?
Electroplating is the process of coating one metal or metal object with a very thin layer of another metal, typically by applying a direct electric current. Some metals are used as a coating, because they give added protection from rust, damage or corrosion.
What metals can be electroplated?
Many types of metals can be electroplated in this process; gold, silver, tin, zinc, copper, chrome, nickel, platinum and lead. In the diagram below; copper (the anode) is being used to coat another metal (the cathode).
Why is electroplated on iron to make cans used for storing food?
Tin is a non-reactive metal whereas iron is a reactive metal. If food is stored in an iron container, it might react with iron and get spoiled. Hence, tin is electroplated on iron to make cans used for storing food. Electroplating tin makes the container non-reactive and look better.
What are 4 uses of electroplating?
Electroplating used in Aesthetics.
Why do you think electroplated jewelleries are in demand?
Electroplated jewelleries are in demand because firstly, they are as shiny and attractive as real jewelleries. They are light-weighted and cost effective. Secondly, one feels free to wear it because of the growing problem of snatching and theft.
What is electroplating in simple words?
The process of coating the surface of a conducting material with a metal. Electroplating is usually used to cover a less expensive metal with a more expensive metal, or to cover a corrosive metal with a less corrosive or noncorrosive metal.
Can stainless steel be electroplated?
Can You Plate Onto Stainless Steel? Yes, plating onto stainless choice is an excellent choice for various applications. SPC can electroplate your stainless steel products using the same techniques we use for electroplating any other parts.
Can aluminum be electroplated?
Electroplated aluminum can be applied over most engineered materials to meet challenging surface finishing requirements and enhance product performance. You can apply Aluminum electroplating on steel, copper, titanium, and more to improve your products.
Which metal is electroplated on iron for making cans used for storing food?
Tin
Tin is a non-reactive metal whereas iron is a reactive metal. If food is stored in an iron container, it might react with iron and get spoiled. Hence, tin is electroplated on iron to make cans used for storing food.
Why is electroplated metal used in electroplating process?
Because metals are inherently conductive, electroplating is a great way to increase the conductivity of a part. Antennas, electrical components, and other parts can be electroplated to increase performance. Though electroplating boasts plenty of benefits, its limitations lie in the complexity and hazardous nature of the process itself.
How many metals can be electroplated on a surface?
Electroplating is the electrodeposition of metals onto a surface. Conventional aqueous electroplating is limited to about 22 metals and a few dozen alloys. Good luck. finishing.com is made possible by …
What kind of metal can be electroplated in a computer?
A. Hi, Nafateeri. There are a half dozen different electroplated metals just in the computer you just typed your question into — including copper, nickel, tin, and gold, and usually some silver, palladium, and chromium. Some of this electroplating is used for carrying current, some is for corrosion resistance, and some for decorative purposes.
How is Electrolysis used to plate a metal?
Engineers use controlled electrolysis to transfer the desired metal coating from an anode (a part containing the metal that will be used as the plating) to a cathode (the part to be plated). Diagram of copper electroplating using an electrolyte bath of copper sulfate, sulfuric acid, and chloride ions. ( image source)
Because metals are inherently conductive, electroplating is a great way to increase the conductivity of a part. Antennas, electrical components, and other parts can be electroplated to increase performance. Though electroplating boasts plenty of benefits, its limitations lie in the complexity and hazardous nature of the process itself.
Electroplating is the electrodeposition of metals onto a surface. Conventional aqueous electroplating is limited to about 22 metals and a few dozen alloys. Good luck. finishing.com is made possible by
A. Hi, Nafateeri. There are a half dozen different electroplated metals just in the computer you just typed your question into — including copper, nickel, tin, and gold, and usually some silver, palladium, and chromium. Some of this electroplating is used for carrying current, some is for corrosion resistance, and some for decorative purposes.
Engineers use controlled electrolysis to transfer the desired metal coating from an anode (a part containing the metal that will be used as the plating) to a cathode (the part to be plated). Diagram of copper electroplating using an electrolyte bath of copper sulfate, sulfuric acid, and chloride ions. ( image source)
Improving properties. One use of electroplating is to improve the resistance of metal objects to corrosion . It also improves their appearance and may be used to produce gold-plated jewellery. the electrolyte contains ions of the plating metal.
What happens in electroplating?
Electroplating is a process that uses an electric current to reduce dissolved metal cations so that they form a thin coherent metal coating on an electrode.
What are the two main reasons for electroplating objects?
Uses of Electroplating
- Aesthetics.
- Commercial applications.
- To prevent corrosion.
- Conduction of Electricity.
- Reduce friction.
- To protect from radiation.
Is electro plated real gold?
Modern gold plating, called gold electroplating, relies on a chemical process to combine various layers of metal into a solid piece with a layer of gold resting on the surface. Modern electroplating begins with the creation of a metal salt using the plating metal, typically gold or silver.
How is electroplating used in everyday life?
Electroplating is used in various commercial appliances. Nickle is used in decorative items, cars, and machinery parts. Chromium is also used in rims of wheels and zinc is also plated on various machinery parts.
What objects are electroplated?
Prepare a list of objects around you that are electroplated
- Utensils of Kitchen.
- Kitchen Stove.
- Water pipe.
- Water Tap.
- Handle bars and wheel rims of Bikes.
- Artificial Jewellery.
- Door Handles.
What is 18K electroplated gold?
18K Gold Electroplated Jewelry is not 18K gold jewelry, but covered with a thick layer of 18k real gold. They look alike in appearance, you can see that 18K Gold Electroplated Jewelry density and hardness are bigger than 18K gold jewelry.
Is electroplated gold worth anything?
If you’re looking to resell your gold plated jewelry item and want to know if it is worth anything, the truth is that gold plated jewelry items are not worth much. The cost to refine the plated item is higher than the cost of a solid gold item (10K to 24K), so there really isn’t any value in refining it.
What is electroplating give two examples electroplated objects?
Pots of metals, bath taps, ornaments, rims of vehicles, handle bar of cycles and motor cycles, kitchen gas burner, bottom of cooking utensils, handles of doors, tin cans are the some objects around us that are electroplated.
What is electroplating prepare a list of object around you that are electroplated?
What metals Cannot be electroplated?
Which of the following metals cannot be electro plated? Explanation: Since tungsten does not have discharge capacity, it cannot transfer electrons from one electrode to another easily which is why it can be electroplated. Tungsten is the least electroplated metal.
Can steel be electroplated with aluminum?
Compatible Materials For Electroplated Aluminum. Electroplated aluminum can be applied over most engineered materials to meet challenging surface finishing requirements and enhance product performance. You can apply Aluminum electroplating on steel, copper, titanium, and more to improve your products.
Can I wear 18K gold everyday?
The alloy is what makes the gold stronger, and is perfect for everyday wear. 18k gold is typically seen as a special occasion gold, such as engagement rings, anniversary rings, etc. Since we started Kinn, we often get asked whether you can wear our jewelry if you’re allergic to nickel.
Is 24K electroplated worth anything?
If it’s only electroplated with gold, it has zero intrinsic metal value and is only worth it’s decorative value as costume jewelry.
What is 18K gold electroplated?
The most common form of electroplating is used for creating coins US pennies, zinc covered with copper, In a same way, 18K gold electroplated is a way to deposing 18K real gold on the surface of less expansive material like copper,brass or stainless steel by electrodeposition.
What does it mean 18K gold electroplated?
When you buy 18 karat gold plated rings, chains, or bracelets, it means that the golden layer which covers it contains 75% of pure gold. Make sure that you understand that the layer is a really thin layer, so the amount of pure gold in it does not drastically increase its value.